|
The cookie is used to store the user consent for the cookies in the category "Analytics". This cookie is set by GDPR Cookie Consent plugin. These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. Clinical Relevance - Electron Chain Inhibitors This means less ATP is produced, in favour of heat. Clinical Relevance - Pathological UncouplingĪdministration of these drugs results in overheating as the protons diffuse through the membrane and not through ATP synthase. The previous stages of respiration generate electron carrier molecules, such as NADH, to be used in the ETC.Ĭlinically, some molecules interfere with the electron transport chain, which can be life-threatening and will be discussed in this article. The electron transport chain (ETC) is the main source of ATP production in the body and is vital for life. Therefore, there is minimal ATP production. This means that all complexes before that remain coupled with an electron, and so there is no passing of electrons down the chain and no proton motive force established. Clinical Relevance – Electron Chain Inhibitorsīy inhibiting a protein in the ETC sequence, the proteins can’t use the energy of the electrons to pump hydrogen ions, and so the chain can’t function.Įxamples include cyanide and carbon monoxide which inhibit the final electron acceptor. This is also seen in salicylate, which is a by-product of aspirin poisoning.Īdministration of these drugs results in overheating as the protons diffuse through the membrane and not through ATP synthase. This is seen in 2,4-dinitrophenol (DNP) poisoning, where the membrane becomes more permeable to protons. In contrast to uncoupling proteins, some chemicals increase the permeability of the inner mitochondrial membrane to protons without a need for a protein. Uncoupling can also occur pathologically. Electrons can leak out of the electron transport chain and can reduce oxygen, which can produce free radicals such as superoxide and hydrogen peroxide.Ĭlinical Relevance – Pathological Uncoupling The electrons, meanwhile, combine with the hydrogen ions and oxygen to form water by complex IV. Conversely, when the concentration of ADP is high, there is a lot of ADP for ATP synthase to use and so more ATP is made. Therefore, there is a natural limitation in periods of high respiration to avoid large amounts of ATP from being produced. When the concentration of ATP rises, there is less ADP for ATP synthase to use. This energy generated by hydrogen ions diffusing back into the matrix via complex V is harnessed, thereby creating ATP from ADP. This is key for both pathological and physiological processes and is discussed in uncoupling.ĪTP synthase allows the proton motive force to be discharged and utilised by the cell. The main route for protons to re-enter the matrix is via ATP synthase, or complex V. This is an electrical and chemical gradient of hydrogen ions between the intermembrane space and the matrix. In doing so, a proton motive force is generated. This process generates energy which is used to pump hydrogen ions into the intermembrane space. These electrons are then passed along to the next complex in the chain. The two electron carriers, NADH and FADH 2, begin the chain by donating their electrons to complex I and complex II respectively. 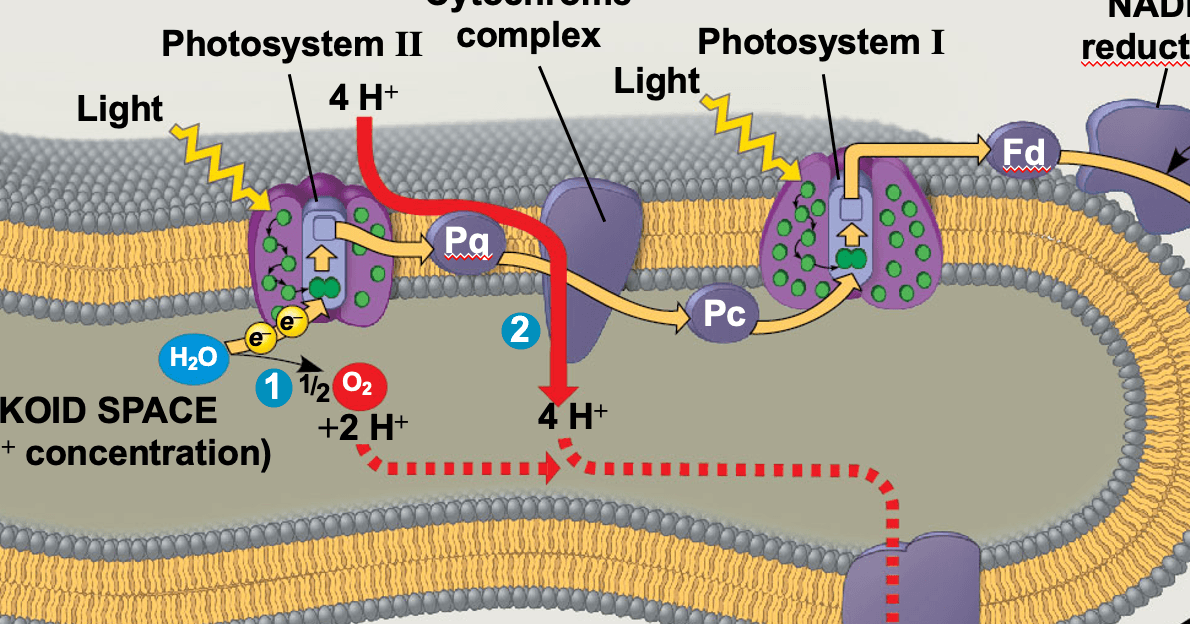 
These are labelled complexes I, II, III, IV, and V. There are five main protein complexes in the ETC, located in the inner membrane of the mitochondria. The electron transport chain is located in the mitochondria.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
